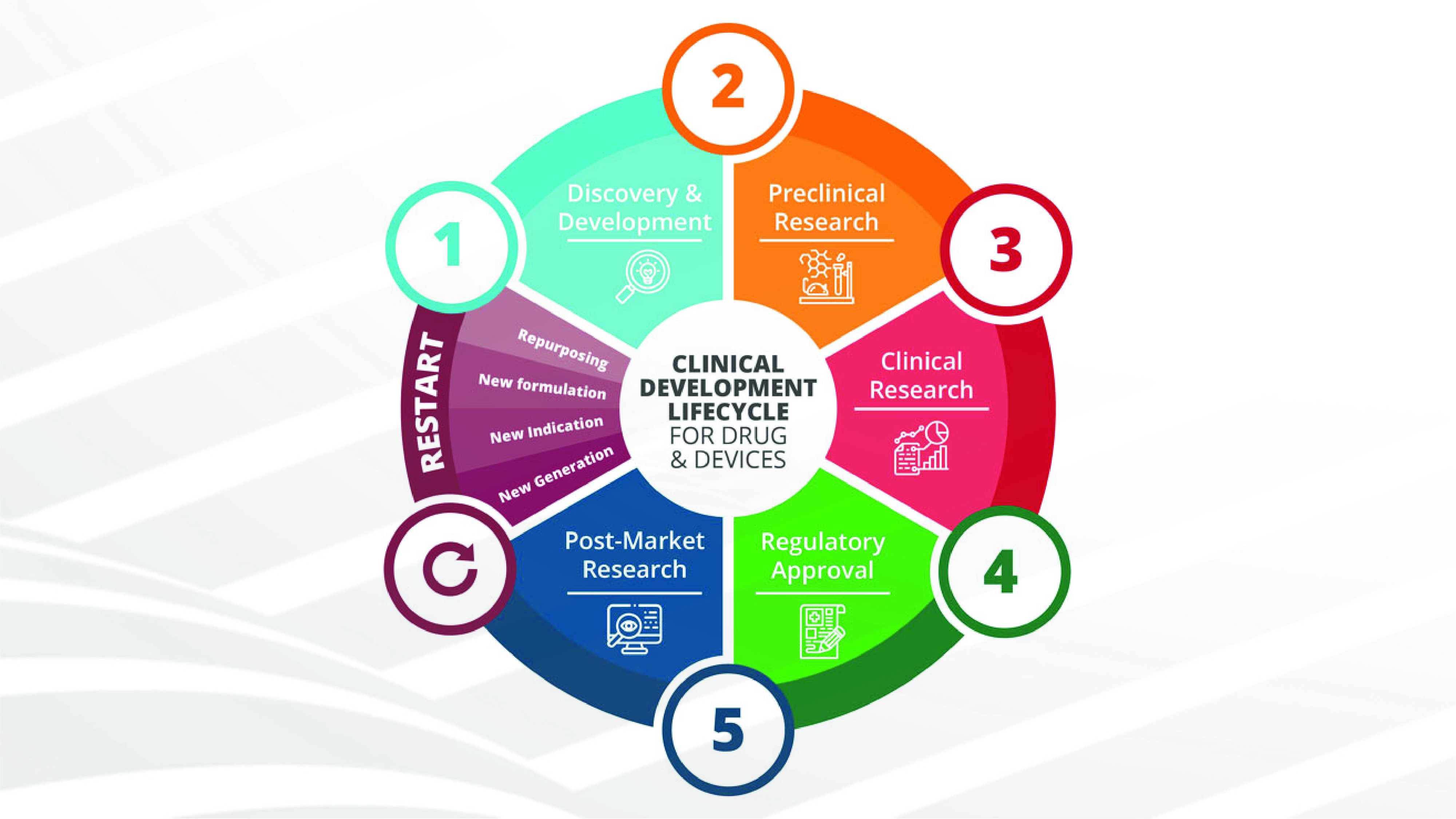
CLINICAL DEVELOPMENT OF DRUGS AND MEDICAL DEVICES: AN OVERVIEW OF THE MOST INNOVATIVE AND INTERESTING ASPECTS.
Clinical development is a term traditionally used to define the entire process of bringing a new drug or medical device to market. It includes a pre-clinical phase to support human use and a clinical trial phase on patients, in compliance [...]