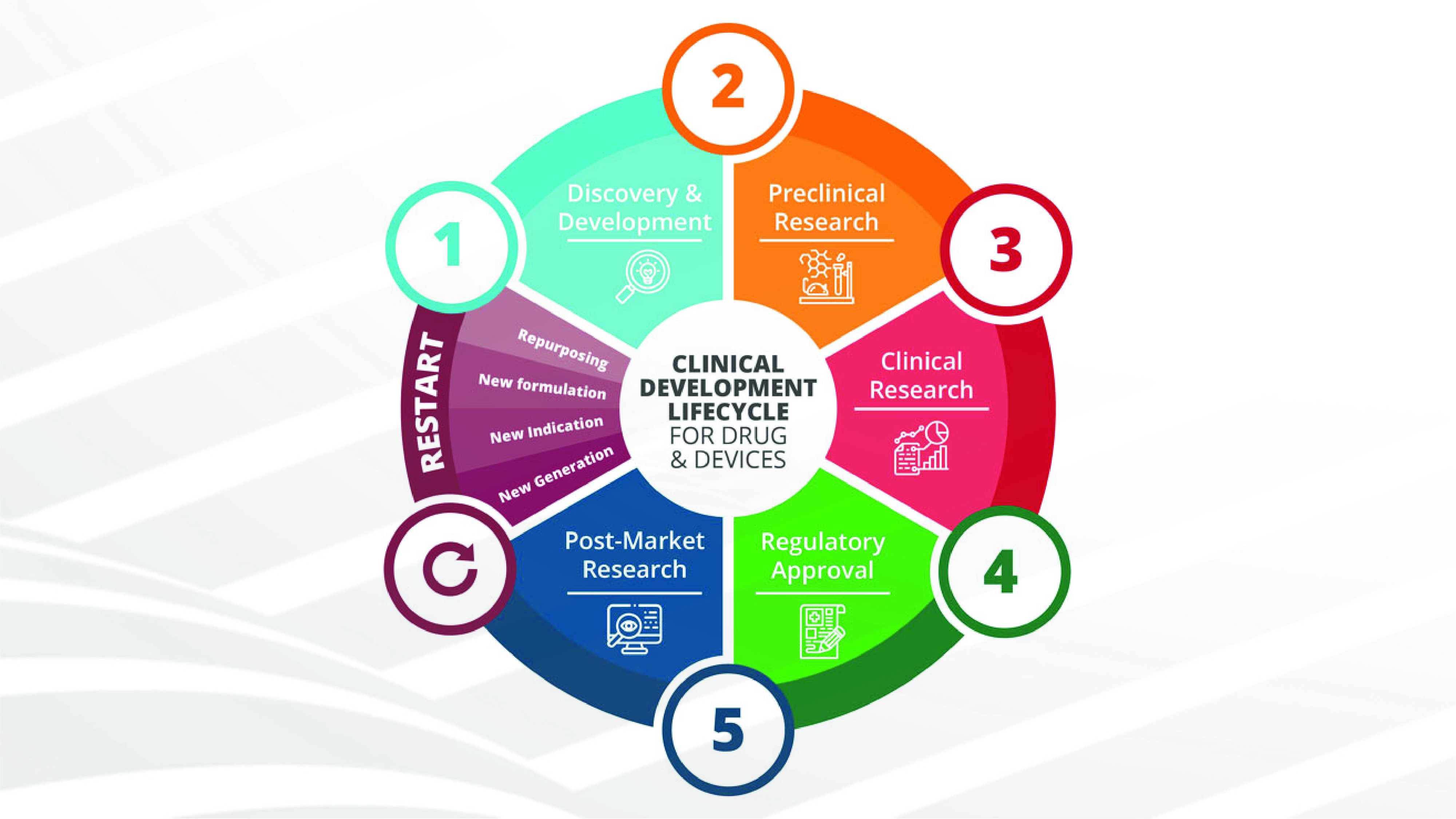
MEDICAL DEVICES AND MDR: SUSTAINABLE SOLUTIONS FOR SURVEILLANCE AND POSTMARKETING STUDIES
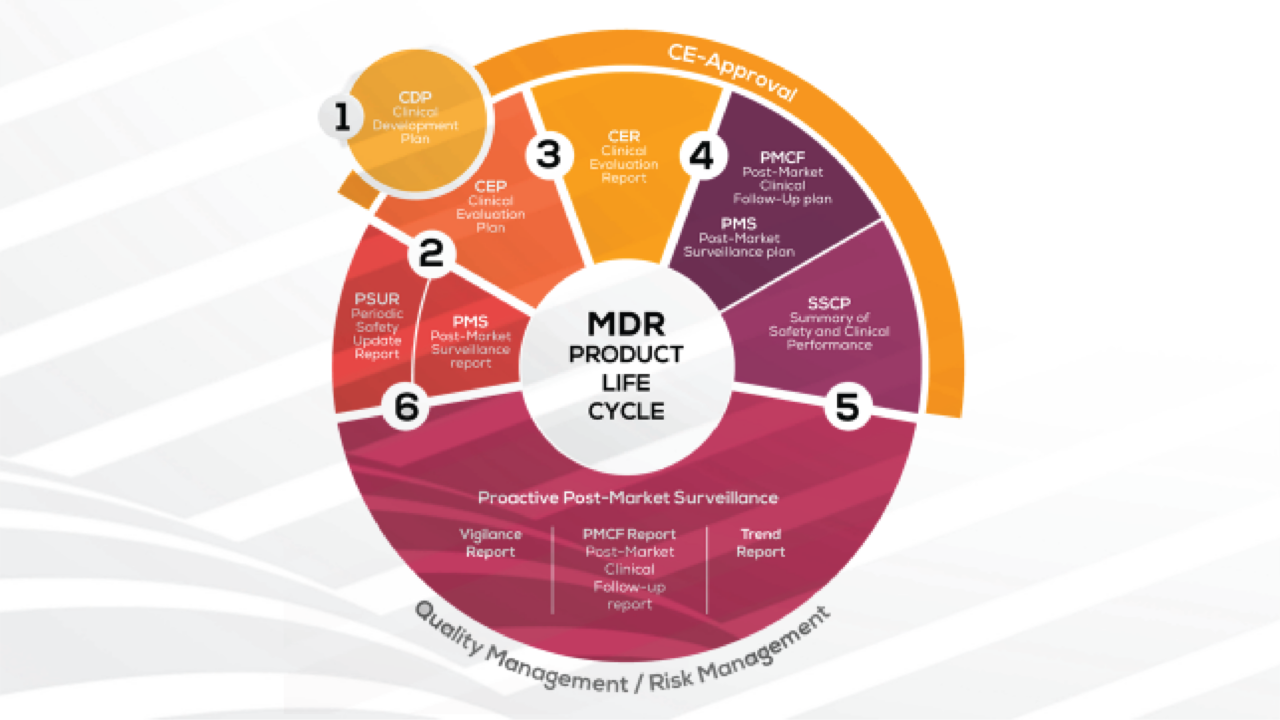
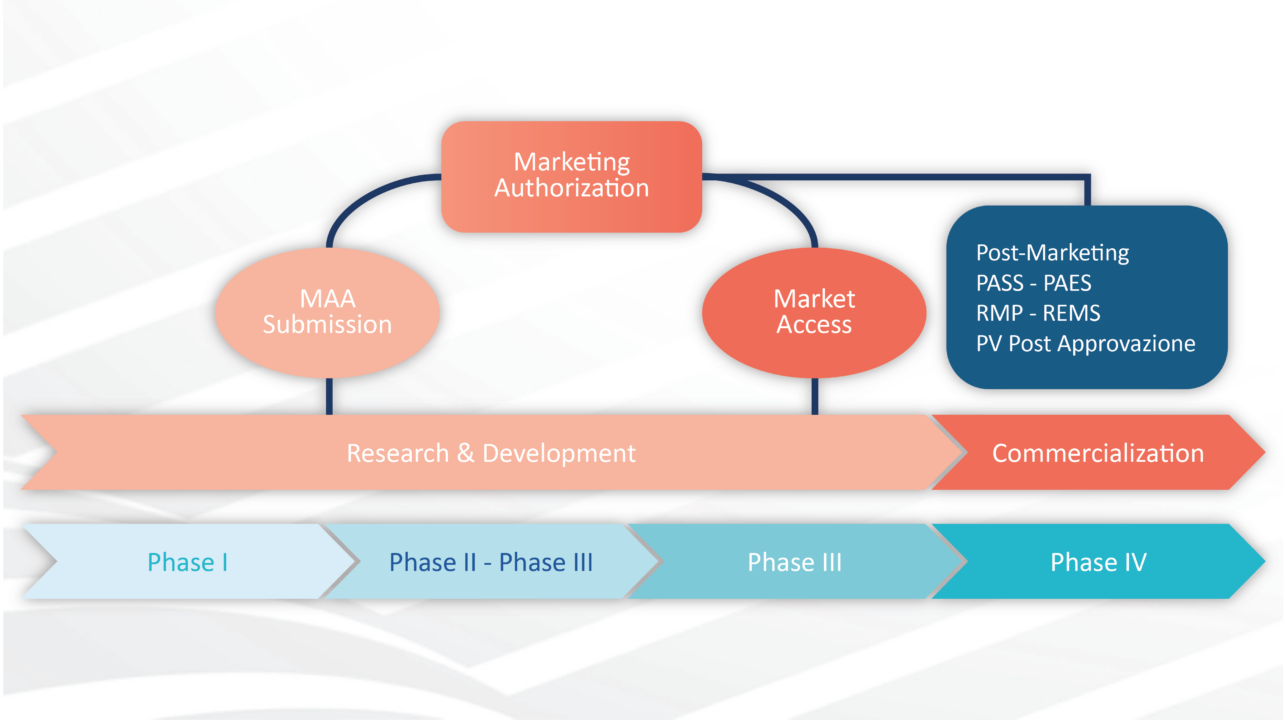
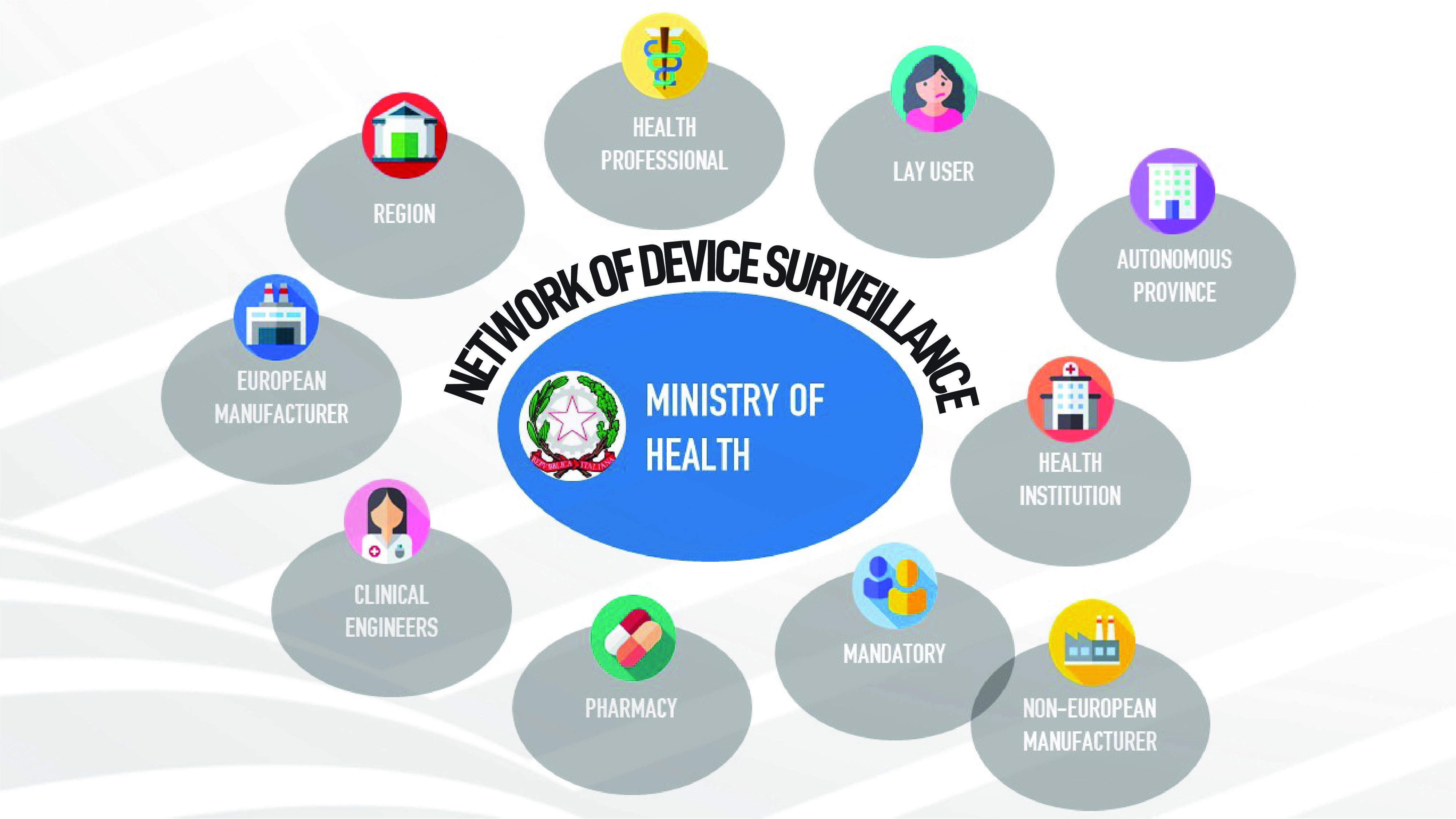
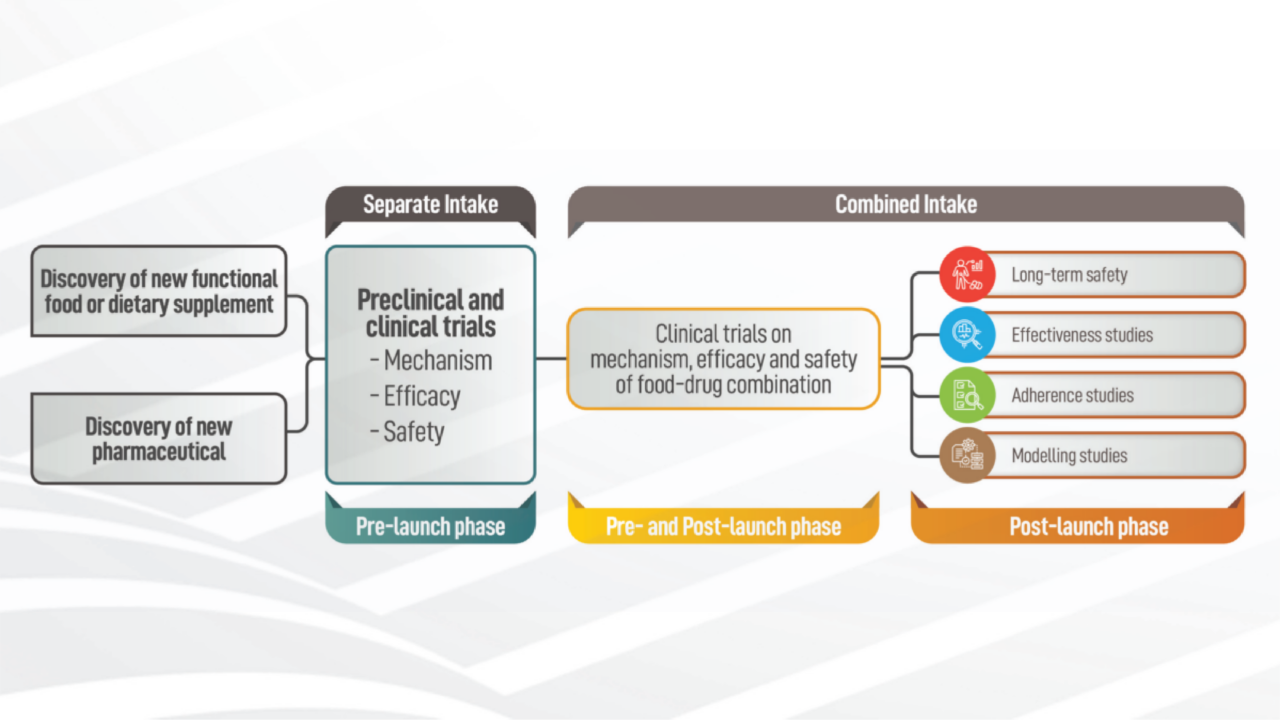
After more than 20 years, new legislation, the "Medical Device Regulation" (EU-MDR 2017/745), has come into force in Europe. The triggers for the initiation and implementation of this European law, which is much stricter than previous directives, were scandals related [...]