ABOUT US.
REGULATION TECHNOLOGY INTEGRATION
INSPIRED BY EXCELLENCE & INNOVATION
The smart choice for your journey through clinical trials.
Meditrial is a leading, full services Clinical Research Organization (CRO) focused on the needs of the medical device industry. We are proud to serve the most innovative life sciences companies in the world.
Meditrial is renowned as the go-to partner by pharma and medical device companies developing breakthrough devices that require a clearly defined strategic path to first-in-man and pivotal clinical studies.
Established in 2008, today we are a trusted Representative for large corporations as well as start-ups requiring support in the ever-changing horizon of the MedTech Standards and Regulations. Despite the increasing difficulty to obtain approval for clinical investigations, we have an outstanding record of 100% success in assisting clients.
Our proprietary cloud-based software solutions are designed to minimize complexity, manage large trial results and ensure the efficiency of medical device or pharma development, launch, and adoption.
We’ll take care of the in-depth, daily clinical operations. You can enjoy the rest of the journey through the MedTech, Pharma and Biotech universe.
You can find us in our headquarters in Zürich, but also in our offices located in Rome, Berlin and New York.
THERAPEUTIC EXPERTISE
Meditrial manages projects in multiple therapeutic areas. Knowledge of your therapy ensures compliance and facilitates execution of your trial.
- Cardiac Valve Repair and Replacement
- Structural Heart Interventions
- Coronary and Peripheral Interventions
- Cardiac Ablation and Pacing Devices
- Neurology and Neurovascular
- Ophthalmology
- Diagnostic Devices
- Orthopedics
- Software Devices
- Wearable monitors
- Oncology
- Endocrinology & Diabetes
- Microsurgery & Robotics
- Female Health & Gynecology
- Dermatology
- Dental care
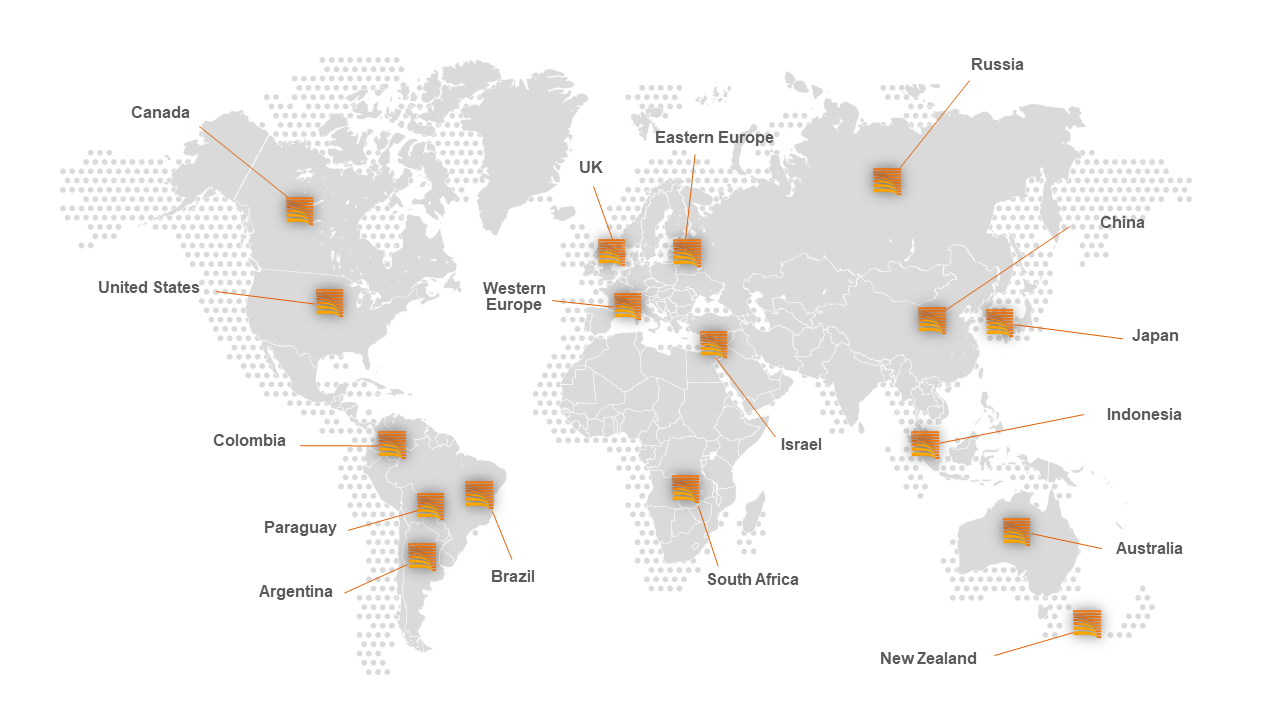
WORLDWIDE CLINICAL TRIALS
Managing projects in other countries can become difficult. If you do not have the right experience and if you do not have the necessary knowledge, the process can become much more complex.
Meditrial has twenty years of experience and is present on all continents, having connections with the main world views. Meditrial manages multiple therapeutic areas, in different countries. Knowledge of your therapy ensures compliance and facilitates execution of your trial.