Paola Antonini, Meditrial Scientific Director.
Due to the pandemic and the information shared on social media, the increased attention of individuals to their own health and well-being has led to a considerable increase in the consumption of food supplements such as vitamins, minerals, immune stimulants, prebiotics and probiotics.
Today, however, constructing a nutraceutical diet is very difficult precisely because the substances actually contained in a food are not known in detail.
Clinical trials are an essential component to substantiate the claims made on food labels, including supplements.
GROWTH OF THE NUTRACEUTICAL SECTOR: MARKET OPPORTUNITIES
Today, the global market for dietary supplements is around EUR 120 billion with an annual growth rate of more than 6%. In this sector, Europe ranks third behind Asia and North America.
The Italian market ranks first in Europe with a 27% share of the European total (Germany 18% – France 8%) and is worth around EUR 4 billion. According to a recent Census research, 32 million Italians use food supplements (Il Sole 24 Ore).
Italians purchase dietary supplements mainly in pharmacies, a volume that alone accounts for 76.3% of the total value of national sales, followed by para-pharmacies and e-commerce, which registers the highest growth rate at +49.4% over the previous year, alongside large-scale distribution in supermarkets and other chains (+12.7%).
This kind of growth is fueled not only by industrial innovation and the development of new products that meet the demand of health-conscious consumers, but also by health claims covering a wide range of wellness components.
In a highly competitive market, conducting clinical studies is indispensable to provide scientific evidence of the cause and effect relationship of expected benefits (Figure 1).
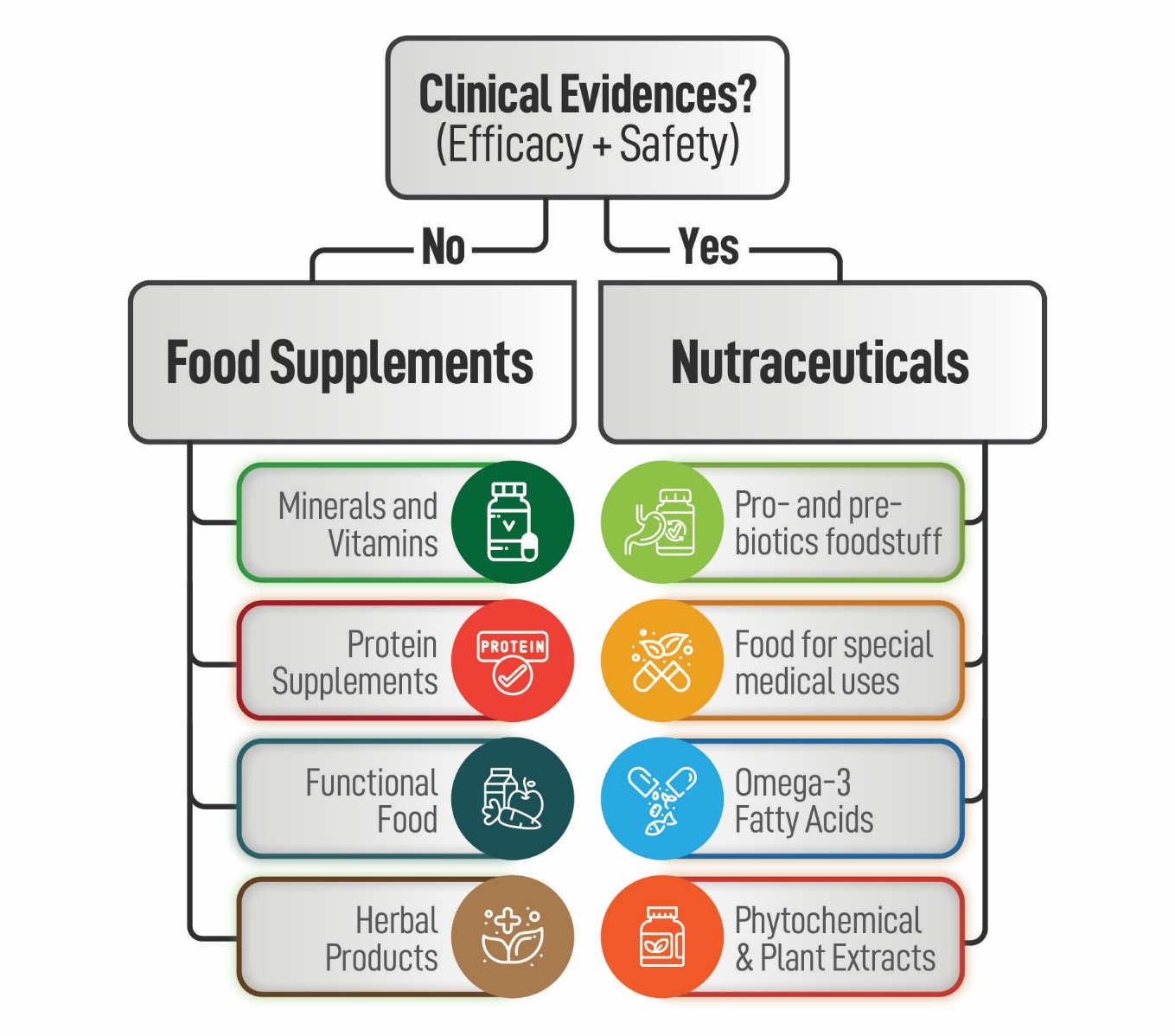
Figure 1. Distinction between dietary supplements and nutraceuticals based on clinical evidence of safety and efficacy
THE NUTRACEUTIC DIET MODEL
“It is becoming increasingly clear how dietary patterns affect health, with significant effects of nutritional components on body functions and disease processes,” emphasizes Paola Antonini, co-author of a wealth of scientific articles on the subject and member of the international research team Ciao (Cilento on ageing outcomes study) with the University of Lund in Sweden and the University of San Diego in California. “The Mediterranean diet, with plenty of fruit and vegetables, olive oil, fish and a moderate intake of wine, has been shown to be associated with a lower risk of both cardiovascular disease and cancer.
The nutraceutical diet is now proposed as a dietary style that, thanks to the healing properties of foods, cures and promotes longevity. But in order to be able to prove the existence of protective effects of supplements or nutraceuticals, it is necessary to carry out specially designed clinical studies, which present considerable complexities since it is necessary to evaluate the benefits and safety of a single food, also in relation to other co-variant lifestyle factors, using a scientific protocol.”
What are the expectations of the public? What are the opportunities and challenges for the development of these products?
SUPPLEMENTS AND HOMEOSTASIS
There is a growing overlap between conventional foods (including beverages) and food supplements.
Food supplements, also called dietary supplements or food supplements, are products intended to supplement the common diet. They constitute a concentrated source of nutrients, such as vitamins and minerals, or other substances with a nutritional or physiological effect, in particular – but not exclusively – amino acids, essential fatty acids, probiotic microorganisms, fibres and extracts of plant origin, both mono- and multi-compound, in pre-dosed forms.
It must be emphasized that food supplements are food products and as such cannot claim therapeutic properties or the ability to prevent or cure diseases (labelling, presentation and advertising) and are subject to food safety regulations. Food supplements are intended to correct nutritional deficiencies, maintain an adequate intake of certain nutrients or assist specific physiological functions. They are not medicines but foodstuffs and, as such, cannot exert a pharmacological action. Therefore, their use is not intended to treat or prevent disease in humans or to modify physiological functions, but to maintain the “homeostatic” balance of a specific function of the body, which has not been deteriorated in a pathological sense, and to demonstrate the reduction of a disease risk factor’, according to the model defined by the Council of Europe (Homeostasis, a model to distinguish between food, including food supplements, and medicinal products, 07/02/2008)
DIFFERENCES BETWEEN FUCNTIONAL FOODS AND NUTRACEUTICALS
“Functional foods”, on the other hand, are ordinary foods to which components or ingredients have been added to achieve a specific medical and physiological benefit, beyond the pure effect of providing nutrition, e.g. milk enriched with Omega-3. “Nutraceutical”, on the other hand, is ‘a food (or part of a food) that provides health benefits’, on the borderline between nutrition and drug therapy.
The difference is often subtle but relevant in at least two respects: the dose and the associated substances. For example, garlic, which may be useful in controlling mild circulatory system disorders, is a functional food, while a pill containing garlic or its active components is a nutraceutical. Grapes and blackcurrants, which could be useful in many degenerative diseases of the cardiovascular system and in cancer prevention – as they contain antioxidant polyphenols such as resveratrol and quercetin – can be considered functional foods, while the polyphenols derived from them are nutraceuticals.
However, to date, the term nutraceutical has no clear regulatory definition. Therefore, nutraceuticals are classified in the category of food supplements regulated by Legislative Decree 2004 No. 169 and at EU level by Directive 2002/46/EC and subsequent Regulations (Regulation (EC) No. 1925/2006 and Regulation (EC) No. 1170/2009). One of the most complex issues is the fact that many substances, mostly of plant origin, are used both as ingredients in supplements and as active ingredients in medicines. At present, there are no unambiguous scientific and regulatory criteria to distinguish the food and pharmaceutical use of a substance and the two fields of application frequently overlap. Caution must be exercised about the real efficacy of functional foods and nutraceuticals. Many data considered reliable are based on experiments or epidemiological evaluations that are not always easy to conduct and often lack control or statistics.
FOOD OF THE FUTURE
There is a growing overlap between conventional foods (including beverages) and food supplements.
Food supplements, also called dietary supplements or food supplements, are products intended to supplement the common diet. They constitute a concentrated source of nutrients, such as vitamins and minerals, or other substances with a nutritional or physiological effect, in particular – but not exclusively – amino acids, essential fatty acids, probiotic microorganisms, fibres and extracts of plant origin, both mono- and multi-compound, in pre-dosed forms.
It must be emphasized that food supplements are food products and as such cannot claim therapeutic properties or the ability to prevent or cure diseases (labelling, presentation and advertising) and are subject to food safety regulations. Food supplements are intended to correct nutritional deficiencies, maintain an adequate intake of certain nutrients or assist specific physiological functions. They are not medicines but foodstuffs and, as such, cannot exert a pharmacological action. Therefore, their use is not intended to treat or prevent disease in humans or to modify physiological functions, but to maintain the “homeostatic” balance of a specific function of the body, which has not been deteriorated in a pathological sense, and to demonstrate the reduction of a disease risk factor’, according to the model defined by the Council of Europe (Homeostasis, a model to distinguish between food, including food supplements, and medicinal products, 07/02/2008)
THE REGULATORY FRAMEWORK
Currently, supplements may be placed on the market if they have as ingredients the substances permitted by the legislation: the vitamins and minerals listed in CE Regulation 1170/2009, the botanicals listed in Annex 1 of the Ministerial Decree of 10 August 2018, probiotics as per the Ministry of Health guidelines, and other nutrients and substances with a nutritional or physiological effect.
Clinical trials are an essential component to substantiate health claims, also known as Health Claims, on food labels, including supplements. When properly conducted, such studies provide the highest level of scientific evidence in a cause and effect relationship.
Clinical trials can therefore activate huge market opportunities for food supplements, providing evidence of health benefits that distinguish the many products on the shelves of pharmacies, prepharmacies and supermarkets. Unfortunately, in the EU there are many generic and widely used health claims, as Article 13.1 of the CE EFSA Regulation allows manufacturers to use a large number of ‘health claims’, including those concerning vitamin and mineral content.
In the United States, health claims on food labels are examined by the FDA, while within the European Union it is the responsibility of the EFSA. The level of scientific evidence required by EFSA, both in terms of the quality of the clinical study design and the significance of the results obtained, is very high.
TRANSPARENCY IN THE FOOD SECTOR
In recent years, consumer attention to the information on product packaging, especially food packaging, has become more acute. To meet this need, the European Union has stepped in, which, with various regulations in favor of food tracking and traceability, has started a path towards transparency of information and which, unwittingly, is turning into a real marketing strategy on the part of manufacturers.
The food must be clearly characterized in terms of its composition, and, when appropriate, its physical and chemical properties, its manufacturing process, as well as its stability and bioavailability, indicating the category to which it belongs (food, food supplement, food for special medical purposes, etc.).
Hence the need to conduct clinical studies dedicated to the food sector. The problem is that such studies can be complex, as large and small companies often find it difficult to identify the most appropriate study design, to recruit a sufficient number of subjects and to achieve the objectives in terms of time and cost of the research, according to Regulation 1924/2006.
CLINICAL DEVELOPMENT: THE NEW ROADMAPS FOR NUTRITION
In recent years, the significant growth of the dietary supplement market and the increase in product innovation have encouraged an increase in clinical trials to support the safety and biomedical validity of this type of product.
In the food field, in vitro and in vivo experiments can be carried out on animal models and humans, as described in the Ministry of Health guideline on studies conducted to evaluate the safety and properties of food products.
“Scientific research on nutrition can be conducted through studies of various types (Figure 2). For example, the observational cohort study called Ciao (Cilento on aging outcomes study) involved several thousand patients and produced very robust results on the importance of dietary factors on major diseases, such as cardiovascular, metabolic, neurodegenerative and oncological, as well as healthy aging,” reiterates Paola Antonini. “However, such research has also highlighted the difficulty in conducting such large trials that require considerable effort for the patients and physicians involved, with the goal of collecting data on the effect of nutritional factors on health.”
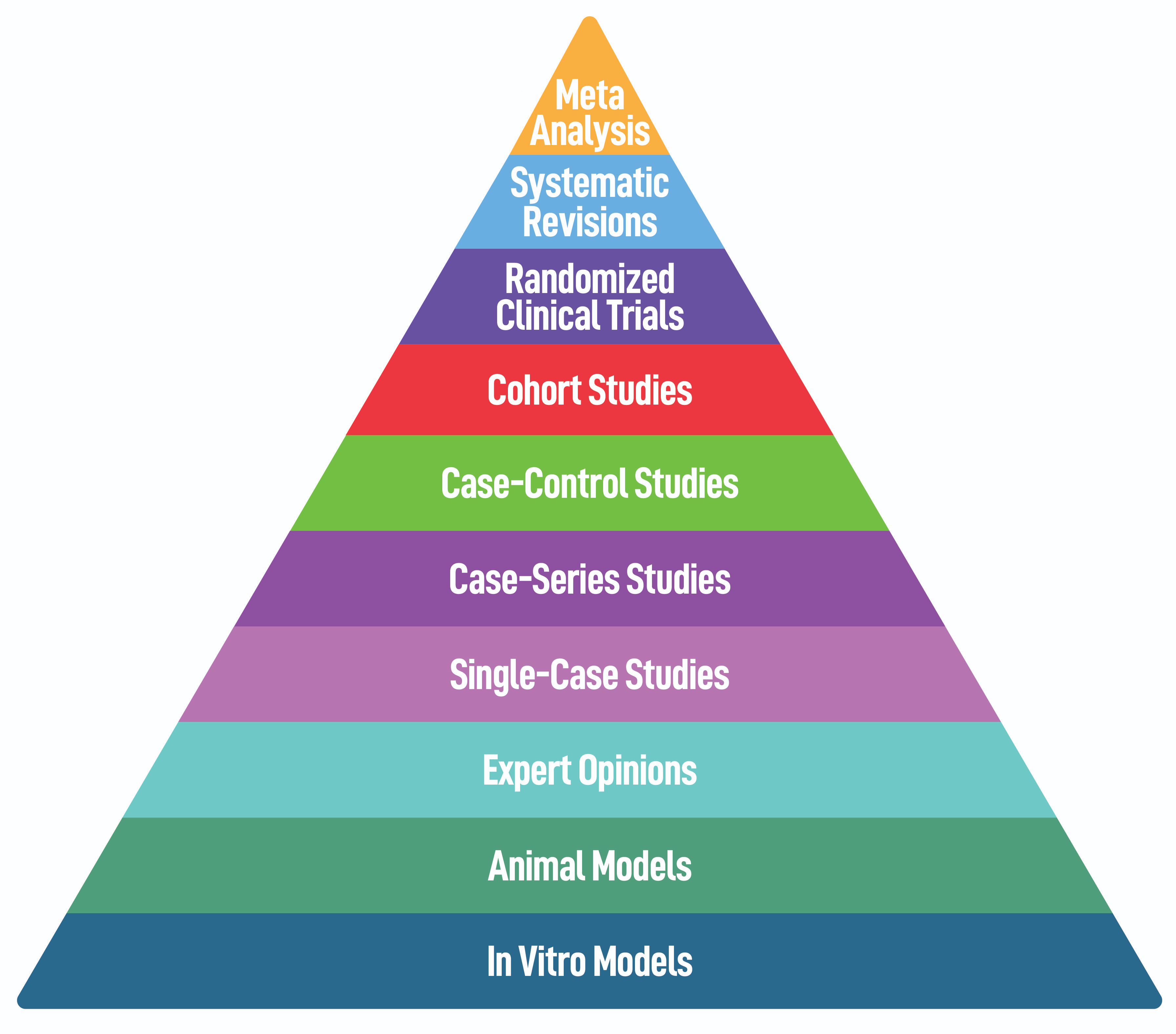
Figure 2. Hierarchy of pre-clinical and clinical trials for studies of dietary supplements and components with nutritional or nutraceutical activities
HOW TO CONDUCT CLINICAL TRIALS FOR DIETARY SUPPLEMENTS
Regulation 1924/2006 unambiguously states how to proceed in approving a claim. Nutraceuticals fall between pharmacology and nutrition science and therefore their efficacy must be proven by rigorous clinical studies. This may create the need to conduct a pilot study to obtain detailed information on the desired end results. In addition, recruiting participants can be difficult, as the study group must lead a healthy lifestyle and participate in various monitoring activities to record related information.
Since the health effects of nutraceuticals are easily influenced by the heterogeneity of subjects as well as environmental and lifestyle factors, a large sample size is usually required, which leads to higher costs. In addition, there is often a need to also evaluate the effects of nutraceuticals in conjunction with drugs used for the same therapeutic indication, resulting in the need to evaluate the synergistic or antagonistic effects of the two products used separately, and then in combination (Figure 3).
The challenges for dietary supplement studies are numerous but can be managed with a well-planned and effective trial design. For example, if a trial is conducted to show that a product has the property of reducing a disease risk factor, a significant amount of data is needed and the study will need to last for a longer period. To avoid this, surrogate endpoints can be adopted that are clear to the consumer. For example, instead of stating that a product “reduces the risk of stroke or heart attack,” the surrogate statement is that a product “reduces levels of harmful cholesterol associated with increased risk of stroke and heart attack.”
Where possible, trials should include a comparison group in which the dietary supplement will be compared with a placebo. People often change behavior when they are monitored for a trial, so without a comparison group it is impossible to know whether the changes observed in people taking the dietary supplement are the result of the product or another factor. Double-blind studies, in which placebos are used, are the gold standard and should be used whenever possible. In open-label studies, participants know the therapy they are taking and often withdraw from the study because they perceive no benefit or have no incentive to continue.
A careful balance needs to be struck so that the study population is sufficiently homogeneous to see an outcome, but it should not have peculiar characteristics such that it cannot be generalized to a larger population. In fact, it is important that the study and the data collected be as generalizable as possible to show that a dietary supplement could have an effect on a larger population. Performing the study in different centers ensures that the sponsors can gain insight into possible different demographic and environmental issues and that the data are more reliable.
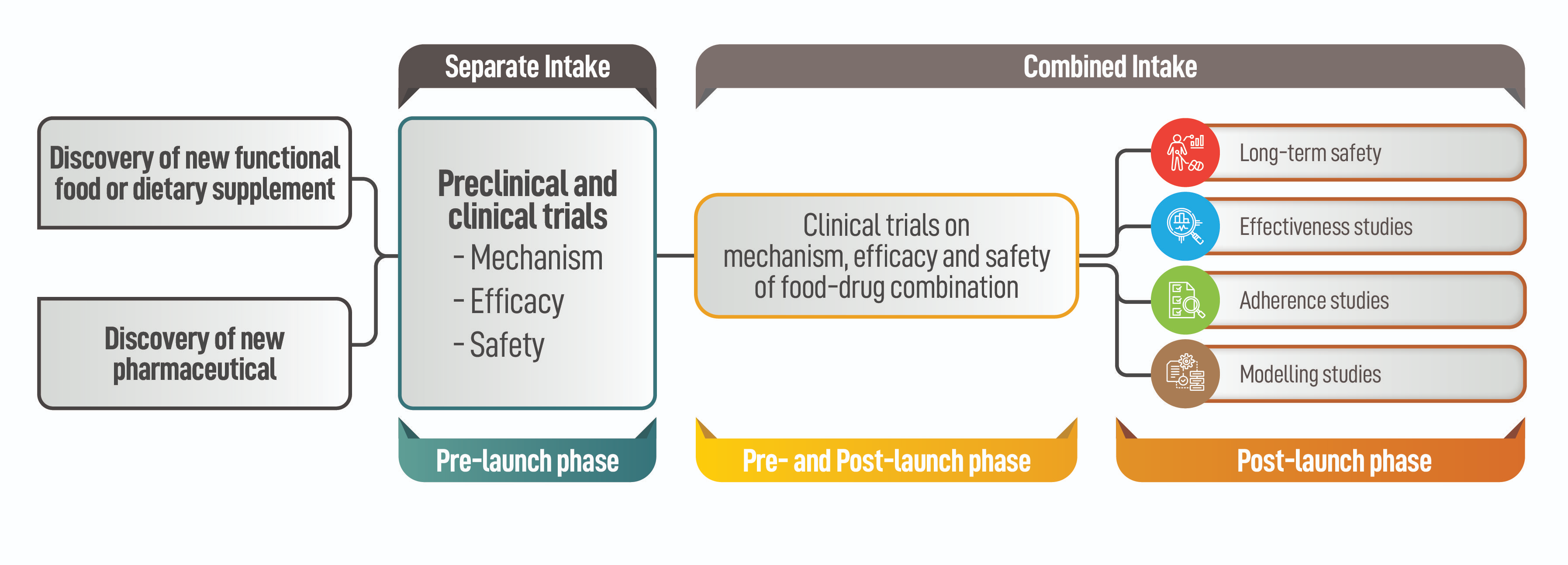
Figure 3. Clinical development of dietary supplements and pharmaceutical products. Separate intake of the two products is necessary to evaluate the mechanism of action, efficacy, and safety. Combined intake is explored in subsequent trials in the pre- and post-marketing phases.
CHALLENGES AND OPPORTUNITIES
Lifestyle choices often have a greater influence than the effects of dietary supplements and a more significant impact on the outcome of the trial. For this reason, it is critical that the study design recognizes lifestyle variables and ensures that these are effectively recorded and taken into account in the final statistical analysis. The evidence-based approach applied to nutraceuticals is particular to that taken in other clinical settings: scientific studies in supplements and nutraceuticals must be considered product-specific, and the outcome of trials is not applicable to other active ingredients on the market.
In this highly dynamic field, it is evident how managing the clinical development of supplements may require extensive cross-disciplinary skills in relation to the many actors involved (manufacturers, researchers, participating subjects, ethics committees and relevant authorities, scientific societies and academia) and an interconnected set of increasingly stringent international regulations and guidelines. In this context, partnership with CROs as specialized clinical research organizations takes on the same strategic importance as it has had to date in the clinical development of drugs and medical devices.







Leave A Comment