The UKCA (UK Conformity Assessed) marking will be the UK product marking that will be used for goods being placed on the market in Great Britain (England, Wales and Scotland) as of January 1st 2021.
Meditrial offers market access and regulatory solutions for devices manufacturers. The Meditrial dedicated team of experts will help you to be compliant with new UKCA requirements and ensure market approval for your device. Contact us today!
The UKCA marking will apply to most products which previously required the CE marking, including medical devices and IVDs. The UKCA will not be recognized on the EU market. Products that require CE marking will still need a CE marking to be sold in the EU.
However:
- CE marking will continue to be used and recognized in Great Britain until 30 June 2023
- Certificates issued by European Economic Area (EEA)-based Notified Bodies will continue to be valid for the Great Britain market until 30 June 2023
If you are a manufacturer based outside the UK and wish to place a device on the UK market, you will need to establish a UK Responsible Person who will take responsibility for the product in the UK.
Learn more about Meditrial Market Access Services.
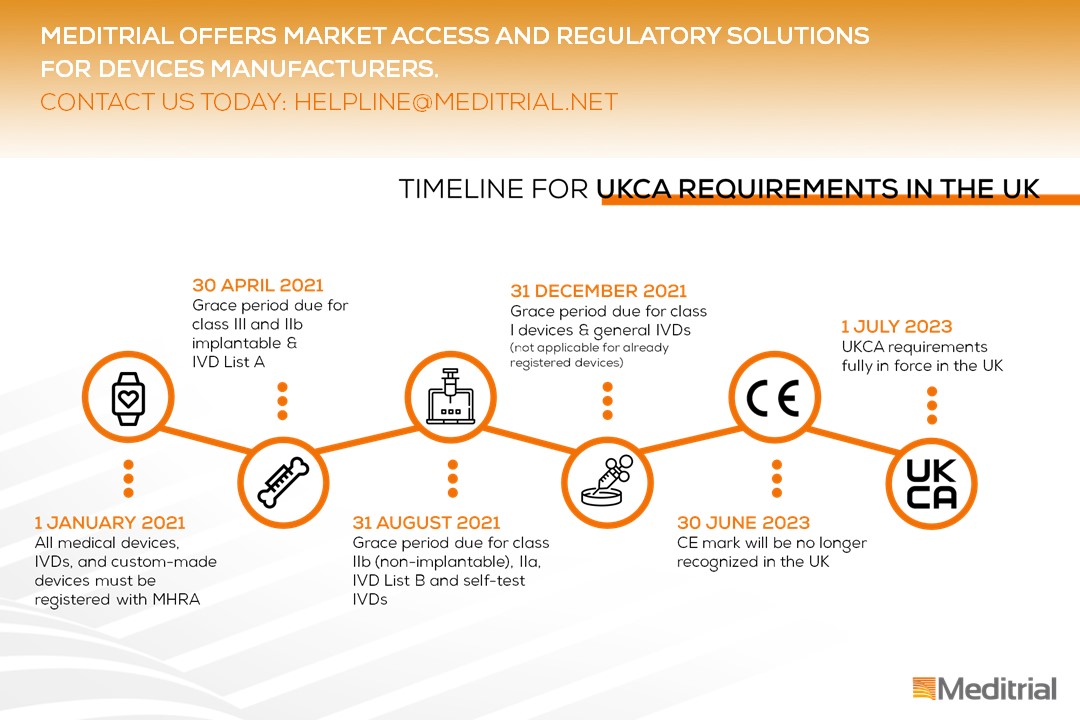
Device registration in the UK as of January 1st 2021
These new registration requirements apply as of 1 January 2021 according to the following timeline:
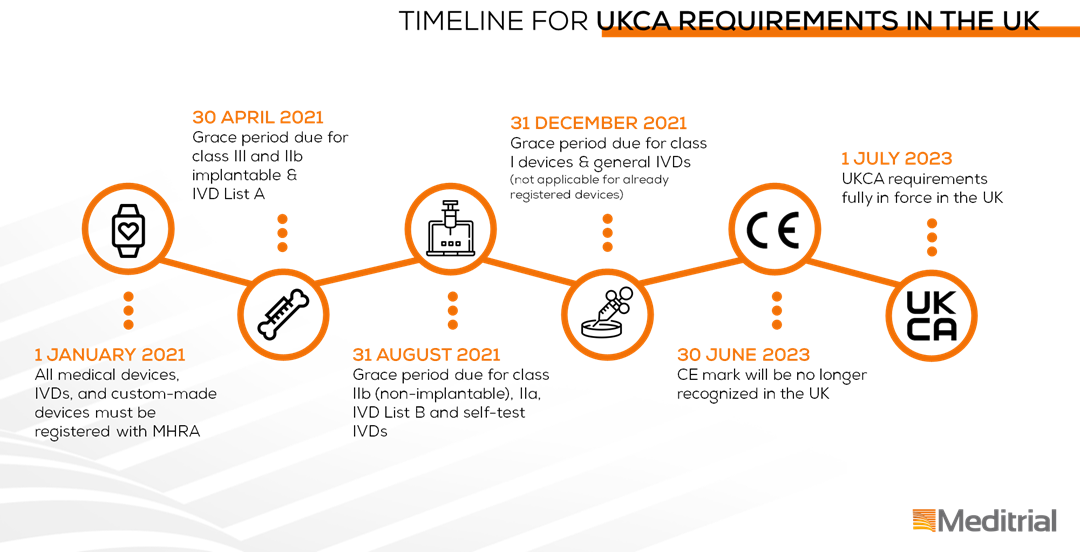
Please note, the information provided are based on the current guidance which might be subject to change as negotiations between the UK Government and the EU continue. Meditrial is constantly observing this situation and will continue to inform you on future developments.
For more details, please see the guidance from the MHRA.
Contact Meditrial for immediate assistance.







Leave A Comment